Activity
Test for Acids or Bases Using...Flowers!
When certain plants come into contact with acids or bases, their colors change! These changing plants are acid-base indicators, and they are generally alkaline or acidic, so when they come into contact with things that have a totally different ph, their colors change.
This hands-on science project will let your child see if your own plants are acid-base indicators!
What You Need:
- At least 3 types of purple, orange, red, blue, or pink flowers
- Safety glasses
- Water
- Permanent marker
- Baking soda
- White vinegar
- Measuring cups
- At least 5 clear plastic cups
- At least 3 spoons
What You Do:
- Put a half cup of vinegar into a plastic cup, and ask your child to label it "acid."
- Put a half cup of water, and mix in a couple teaspoons of baking soda. Label this cup "base."
- Invite your young scientist to pick a flower and crush up petals into tiny bits. Separate these into three cups, and mix a few drops of water into each.
- Take a couple drops of acid with a spoon, and add it to one cup. Do the same with the base in another cup. What happens? How do these compare to the third flower that didn't have an acid or base added to it?
- Acid-base indicators will change quickly! Ask your child if she thinks the flower is an acid-base indiciator. After contact with a base, a red flower will turn blue or greenish. After contact with an acid, a red flower will get more red. Blue flowers, after contact with a base, will turn more blue, and after contact with an acid, will become more pinkish.
- Try steps 3 and 4 with the other flowers. Which flowers are acid-base indicators? To get accurate results, clean the flower cups to make sure no leftover residue remains.
- Challenge your scientist to reverse the reaction! Is it possible to add more of an acid or base to return a flower to its original color?
Not all flowers are acid-base indicators. Tulips, pansies, and roses are just a few of many flowers that are acid-base indiciators. If you need more options, take some fruit and vegetables from your refrigerator to test!
Related learning resources

Watch Acids and Bases Produce Colors!
Activity
Watch Acids and Bases Produce Colors!
Show your child how to concoct a simple duo of household ingredients that changes color as acids and bases are added!
5th Grade
Science
Activity
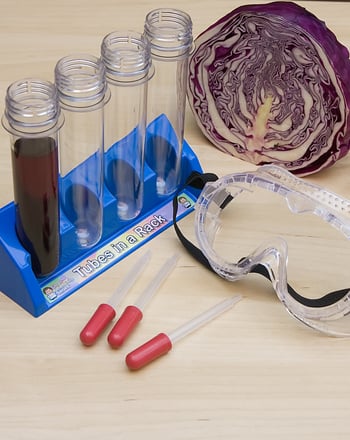
The Red Cabbage Acid Test
Activity
The Red Cabbage Acid Test
How to test for acids and bases? In this activity you and your child can make your own indicator from red cabbage!
5th Grade
Science
Activity

Five Tips for Calmer Test-Taking
Worksheet
Five Tips for Calmer Test-Taking
Stay calm and centered during your next quiz or exam with these test-taking tips!
1st Grade
Worksheet

Egg Carton Flowers
Activity
Egg Carton Flowers
Use these pretty egg carton flowers to decorate a spring table. They don't require water, they'll never wilt, and they're perfect for Easter.
Activity

Duct Tape Flowers
Activity
Duct Tape Flowers
Make a bouquet of colorful duct tape flowers in this hands-on spring craft.
Activity

Flowers Everywhere: Practice Coordinates
Worksheet
Flowers Everywhere: Practice Coordinates
Challenge your child to help Emma put together a beautiful bouquet by tracking down the coordinates for a bunch of flowers.
Worksheet

Make Corn Husk Flowers
Activity
Make Corn Husk Flowers
This easy craft project shows you and your kid how to make pretty corn husk flowers using just a few tools.
4th Grade
Activity

Streak Test
Activity
Streak Test
In this science fair project, students use the streak test to tell minerals apart from each other.
2nd Grade
Activity

How to Press Flowers
Activity
How to Press Flowers
April showers may bring May flowers, but your child can preserve the beauty of spring's bounty all year long with this simple craft project!
1st Grade
Science
Activity

Tensile Strength Test
Activity
Tensile Strength Test
In this science experiment, do tensile strength test on fishing line, a strand of hair, a rubber band, and any other materials you're curious about!
5th Grade
Activity

Ikebana: Flower Arrangement
Activity
Ikebana: Flower Arrangement
Learn about Ikebana, or the Japanese art of flower arranging, with your child while artfully arranging spring flowers!
5th Grade
Science
Activity

High Frequency Hearing Test
Activity
High Frequency Hearing Test
Little scientists will make inferences about hearing loss, pitch, and frequency in this great science fair idea that puts the human ear to the test.
Science
Activity



