Activity
Air Pressure Science Experiment: Balloon and a Jar
Grade Level: Kindergarten to 2nd (with adult supervision!); Type: Physics
Objective:
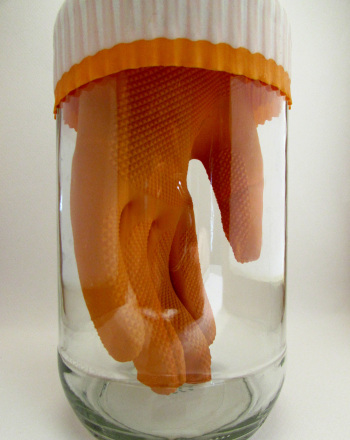
Use heat to change the air pressure inside a bottle causing a balloon set on top of the bottle to squeeze into it.
Research Questions:
When you first set the water balloon on top of the jar, the air pressure outside the jar and the air pressure inside the jar are the same. As the fire burns, air, heated by the fire. rises and pushes its way out of the jar. This hot air pushing out past the balloon causes the balloon to wiggle. However, while hot air is going out, no new air is coming in (the balloon works like a one-way valve, letting air out but not in). So now there is less air, less stuff, in the jar. With less stuff taking up space in the jar, the pressure inside the jar is less than it used to be, that is, less than the air pressure outside the jar. Since the pressure outside the jar is now greater than the pressure inside the jar, what will happen to the balloon?
Materials:
- One large glass jar (a big maraschino cherry jar from a bar works well)
- One balloon
- Water
- A bit of paper
- Matches
Experimental Procedure:
- Fill the balloon with water until it’s a little too big to fit through the mouth of the jar.
- Light a bit of paper on fire and drop it into the jar.
- Quickly place the balloon on top of the jar.
- Observe carefully.
Terms & Concepts: Air Pressure
Education.com provides the Science Fair Project Ideas for informational purposes only. Education.com does not make any guarantee or representation regarding the Science Fair Project Ideas and is not responsible or liable for any loss or damage, directly or indirectly, caused by your use of such information. By accessing the Science Fair Project Ideas, you waive and renounce any claims against Education.com that arise thereof. In addition, your access to Education.com's website and Science Fair Project Ideas is covered by Education.com's Privacy Policy and site Terms of Use, which include limitations on Education.com's liability.
Warning is hereby given that not all Project Ideas are appropriate for all individuals or in all circumstances. Implementation of any Science Project Idea should be undertaken only in appropriate settings and with appropriate parental or other supervision. Reading and following the safety precautions of all materials used in a project is the sole responsibility of each individual. For further information, consult your state's handbook of Science Safety.