Activity
Tap Water, Bottled Water
The pH of the water is an indicator of the hydrogen ion concentration present in it. A pH of 0 means the solution is a strong acid; a pH of 14 means it is a strong base. If the pH is above 7.5, the water may have a poor taste.
High nitrate concentrations may be caused by leaking septic tanks, leaching from landfills, agricultural activity, or leaks from hazardous waste sites or underground storage facilities. Consumption of large amounts of nitrates may be hazardous to human health.
Nitrites are used in water treatment systems as corrosion inhibitors, as preservatives, and in the manufacture of organic chemicals. Ingesting nitrites repeatedly may be linked to increased incidences of cancer.
The hardness of water is a measure the calcium and magnesium concentrations in the sample. High water hardness can contribute to the formation of kidney stones.
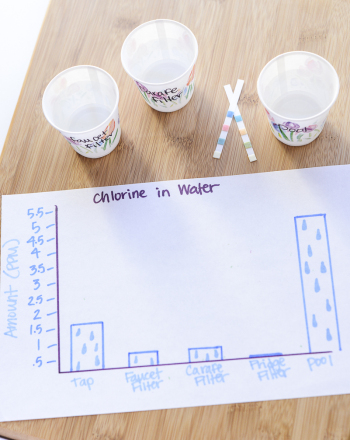
Chlorine is used to disinfect municipal water supplies. It may intensify the odor and taste of organic compounds present in the water. Chlorine is also used in higher concentrations as a raw material in the manufacture of chemical products. Free chlorine is the unreacted chlorine present in the water sample; total chlorine is the sum of free chlorine and reacted chlorine.
A primary source of iron in drinking water is aging and corroding water pipes. The consumption of iron in drinking water is not usually associated with adverse health effects. However it can produce undesirable tastes and odors.
The primary source of copper in drinking water is corroding pipes. Elevated levels in copper can cause serious health problems.
Problem:
Are there significant differences between the concentrations of impurities in tap and bottled water?
Materials:
- Tap water
- Bottled water
- Water test kit (The kit should test for pH, nitrate and nitrite concentrations, total hardness, free chlorine, total chlorine, iron concentrations, and copper concentrations.)
Procedure:
- Place some tap water in a clean drinking glass.
- Fill a second clean drinking glass with bottled water.
- Taste the water in each of the glasses. Based on your taste test, formulate a hypothesis about how the two sources of water differ.
- Following the instructions given by the test strip manufacturer, evaluate the tap water for each of the properties that the test strips are designed to evaluate. Record the results for each of the evaluations.
- Evaluate the bottled water in the same way. Record the separate evaluations.
- Compare the results for the two water samples.
- Evaluate your hypothesis in light of the water sample tests. Revise the hypothesis if necessary, and suggest additional tests to check it.
Education.com provides the Science Fair Project Ideas for informational purposes only. Education.com does not make any guarantee or representation regarding the Science Fair Project Ideas and is not responsible or liable for any loss or damage, directly or indirectly, caused by your use of such information. By accessing the Science Fair Project Ideas, you waive and renounce any claims against Education.com that arise thereof. In addition, your access to Education.com's website and Science Fair Project Ideas is covered by Education.com's Privacy Policy and site Terms of Use, which include limitations on Education.com's liability.
Warning is hereby given that not all Project Ideas are appropriate for all individuals or in all circumstances. Implementation of any Science Project Idea should be undertaken only in appropriate settings and with appropriate parental or other supervision. Reading and following the safety precautions of all materials used in a project is the sole responsibility of each individual. For further information, consult your state's handbook of Science Safety.