Activity
Candle Lit Energy
Grade Level: 7th - 9th; Type: Chemistry
Objective:
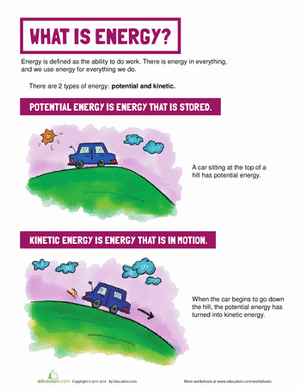
During the burning process, potential energy in a candle is converted to heat energy. This science project measures the amount of heat energy released by a burning candle.
Research Questions:
- How much energy does a candle release when it burns?
- How much potential energy does a candle have?
In order to start a chemical reaction, you need to use some activation energy, such as a burning flame. The activation energy consists of potential energy being converted to a different type of energy, such as kinetic or heat energy. In this project, you can calculate the amount of calories of heat energy that a given amount of candle produces.
Materials:
- Hole puncher
- Juice can
- Stick or glass rod
- Water
- Large can
- Can opener
- Candle stub
- Scale
- Tin foil
- Thermometer
- Match
Experimental Procedure:
- Punch two holes, one in either side of the juice can, near the top. Thread a stick or glass rod through the holes. This will hold the juice can above the larger can.
- Add 100 ml of tap water to the juice can. Leave the water for a few minutes so that it reaches room temperature.
- Remove the bottom of the larger can, so that it is shaped like a metal tube.
- Cut five triangular holes in the bottom of the tube, spaced as equally as possible around the tube. These holes will let air circulate to the candle so that it will continue to burn.
- Weigh the candle stub, and record its weight in grams.
- Stand the tube up, and place a candle on a piece of foil in the center of the tube.
- Use a thermometer to measure the temperature of the water in the juice can. Write down this temperature.
- Light the candle, and immediately place the juice can into the larger can. The juice can should by suspended by the stick or glass rod.
- Stir the water in the juice can with a thermometer until it reads ten degrees higher than the original water temperature. Then blow out the candle.
- Weigh the candle and record the measurement.
- Calculate how many calories of heat were delivered to the water. To do this multiply the mass of the water (100) by the number of degrees the temperature was raised (25). Then divide the product by the number of grams the candle lost during the burning process. This will show you how many calories each gram of candle released, and therefore how much potential energy each gram of candle has.
Terms/Concepts: Potential energy; Calories of heat; Law of conservation of energy
References:
- Easy Genius Science Projects with Chemistry, by Robert Gardener. Pp 65-67.
Education.com provides the Science Fair Project Ideas for informational purposes only. Education.com does not make any guarantee or representation regarding the Science Fair Project Ideas and is not responsible or liable for any loss or damage, directly or indirectly, caused by your use of such information. By accessing the Science Fair Project Ideas, you waive and renounce any claims against Education.com that arise thereof. In addition, your access to Education.com's website and Science Fair Project Ideas is covered by Education.com's Privacy Policy and site Terms of Use, which include limitations on Education.com's liability.
Warning is hereby given that not all Project Ideas are appropriate for all individuals or in all circumstances. Implementation of any Science Project Idea should be undertaken only in appropriate settings and with appropriate parental or other supervision. Reading and following the safety precautions of all materials used in a project is the sole responsibility of each individual. For further information, consult your state's handbook of Science Safety.