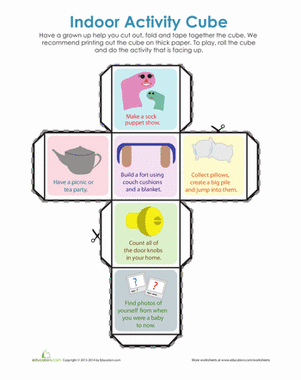
Activity
Catalase Enzyme Activity
Grade Level: 7th - 12th; Type: Life Science
Objective:
Student tests the effects of temperature change on the reactivity of the catalase enzyme.
Research Questions:
- What is a catalyst?
- What is an enzyme?
- How do enzymes work?
- Why is an enzyme’s structure important for it to function?
- How can changes in temperature alter the structure of an enzyme?
- What role do enzymes play in the body?
Enzymes are proteins that act as biological catalysts. They help chemical reactions proceed more easily in the bodies of organisms. Hydrogen peroxide is produced naturally in organisms, but is very harmful. Catalase and other enzymes help break hydrogen peroxide into water and oxygen gas. The bubbling that you may see if you pour hydrogen peroxide on a cut is the oxygen gas produced from catalase activity. Enzymes, including catalase, have very specific structures that help them bind with their substrate and complete the chemical reaction. Changes in temperature and other environmental conditions can alter the structure of a protein. If an enzyme changes its structure, it may no longer work. In this experiment, the student will work to determine the temperature at which catalase denatures and is no longer active. Raw potatoes and liver will act as the tissue samples. Catalase activity will be estimated based on the amount of oxygen bubbles produced.
Materials:
- 1 package raw beef or chicken liver
- 2-3 large raw baking potatoes
- 1 bottle of hydrogen peroxide
- 5-10 small plastic or glass dishes or plastic bags
- 5-10 test tubes or small, clear cups or glasses of the same size
- A knife and cutting boards
- A hotplate or stove
- Pot of water
- Thermometer reading degrees Celsius
- Slotted spoon or fork
Experimental Procedure:
- Create your hypothesis stating at which temperature you believe catalase will be denatured and why.
- Cut liver and potatoes into very small ¼ inch pieces. Cut them on separate cutting boards and be sure to clean the knife between the two. Place potatoes into the pot and just cover with cold water. Keep the liver aside for later. Wash your hands and any surfaces or tools you are finished with.
- Place thermometer into the water with the potatoes. Record the temperature. With a spoon or fork, remove three pieces of potato and place it into a small dish or plastic bag. Write the temperature at which you removed the potato pieces on the dish or bag. Do not seal the dish or bag. Place the pot of potatoes and water onto the stove and turn on low. After the temperature has risen another 5 degrees, remove another three pieces of potato and place into a dish or bag. Record the temperature in your notes and on the dish or bag. Continue heating and removing pieces of potato every 5 degrees. Scientifically, you should do this in degrees Celsius, but if you only have a Fahrenheit thermometer, it will also be fine. With a Fahrenheit thermometer, you may want to remove potatoes only every 10 or so degrees. Whichever you do, be sure to record every temperature as accurately as possible. Stop removing potatoes when they are completely soft, when the water boils, or when the potato is gone, whichever occurs first.
- Set up test tubes or small glasses with equal amounts of hydrogen peroxide in each. If using test tubes, fill about 1/3 full. If using small glasses, use about 5 tablespoons, or fill about an inch deep. Record the measurement of hydrogen peroxide used. Place dishes or bags of potatoes behind test tubes or glasses in the order they were removed from the pot of water. Carefully add first potato pieces removed into the first test tube or glass of hydrogen peroxide. Observe the bubbling and record the amount. If there are lots of bubbles, use ++++, if there are some, use +++, if there are a few, use ++, just a couple, use +. If there are no bubbles, record a 0 for the sample. Continue testing potato pieces in fresh hydrogen peroxide until you have tested them all. Be sure to wash any utensils that touch another sample before using them again. Repeat the entire procedure for the liver.
- Create a line graph of the bubbling activity for both the potatoes and liver. Draw conclusions about the temperature at which catalase is denatured based on your graph. Compare your results to your hypothesis.
Terms/Concepts: Enzymes; Catalase; Hydrogen peroxide; Lock and key mechanism; Substrate; Catalyst
References:
Education.com provides the Science Fair Project Ideas for informational purposes only. Education.com does not make any guarantee or representation regarding the Science Fair Project Ideas and is not responsible or liable for any loss or damage, directly or indirectly, caused by your use of such information. By accessing the Science Fair Project Ideas, you waive and renounce any claims against Education.com that arise thereof. In addition, your access to Education.com's website and Science Fair Project Ideas is covered by Education.com's Privacy Policy and site Terms of Use, which include limitations on Education.com's liability.
Warning is hereby given that not all Project Ideas are appropriate for all individuals or in all circumstances. Implementation of any Science Project Idea should be undertaken only in appropriate settings and with appropriate parental or other supervision. Reading and following the safety precautions of all materials used in a project is the sole responsibility of each individual. For further information, consult your state's handbook of Science Safety.