Activity
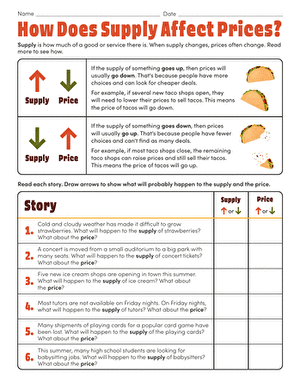
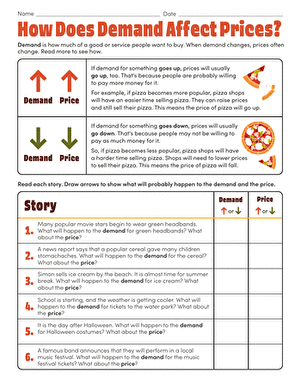
How Do Oil Spills Affect the Environment?
You've heard all about oil spills, but can they really be that bad? After all, olive oil is delicious with Italian food and vegetable oil is in almost every yummy baking good from cookies to brownies. Construct your own ocean habitat to find out for yourself. Next time you hear somehow ask how do oil spills affect the environment, you'll have an answer!
Problem:
How does oil affect an ocean environment?
Materials:
- Cooking oil
- Food dye
- Deep tray
- Water
- Clay
- Moss
- Dish soap
- Handheld fan
- Sponge
- Spatula
- Pencil
- Paper
Procedure:
- Put the clay in your deep tray. The tray will be your "ocean habitat", and the clay will be the land.
- Use the clay to make a shoreline on one end of the tray. Press the sides up tight against the side of the tray. Remember that no shore is perfectly straight -- make sure your shore has curves, bays and inlets.
- Make tiny islands and stick them to the tray's bottom.
- Stick the moss on top of the clay shore and clay islands to represent the plant life.
- Carefully pour water into the tray. Don't drown the islands or the shore! Put just enough water in so that the clay land is still visible above "sea level."
- You're just about ready to add the oil! Before you do that though, think about what might happen when the oil spills into your newly created habitat. Will the oil mix with the water or stay separate? How will the oil interact with the moss and clay land? Write down your thoughts in your notebook.
- Using your notes, take a stab at answering this project's question: how do oil spills affect the environment? Write down your best guess, or hypothesis, in your notebook.
- Pour several drops of oil on one side of the tray, away from your shoreline.
- Draw what the oil does in your notebook.
- Place a fan next to your tray.
- Turn the fan on and direct it at the oil.
- What happens? Draw the oil's movement in your notebook.
- Use a spatula to mimic an ocean's current. Currents move within the ocean. Place the tip of the spatula under the water and push the oil and water mixture together.
- Observe the results and draw it in your notebook.
- Keep adding more oil and spreading it throughout your ocean habitat with the fan and the spatula. Record what happens.
Results:
The oil does not mix with the water. The fan and the spatula should succeed in moving the oil through the water, but the two liquids will never fully combine.
Why?
Water is made up of polar molecules, but oil is made up of nonpolar molecules. Polar molecules have a strong positive and negative charge. Have you ever replaced the batteries in a game controller or a flashlight? The minus and plus sides of a battery show this same sort of positive and negative charge. Since oil molecules do not have a strong positive or negative charge, they can't stick with the water molecules. It doesn't matter how much you may have tried to stir and mix the liquids together -- oil and water will never mix because of their polarity.
Think about how your experiment relates to the real world. Now you know that when oil accidentally spills in the ocean, the water and the oil do not mix. Can you think of how this might be bad for the plant and animal life that rely on clean ocean water? If oil can not dissipate, or spread evenly, throughout ocean environments, then some plants and animals will be stuck in the thick mixture without a way to survive.
Keep experimenting with your ocean habitat! Scientists still do not know the best way to fix oil spills. Guess and test different ways to remove the oil from your habitat. See what dish soap does when you add it to the mixture or even a giant sponge. Can you find the best method to get rid of the oil?
Education.com provides the Science Fair Project Ideas for informational purposes only. Education.com does not make any guarantee or representation regarding the Science Fair Project Ideas and is not responsible or liable for any loss or damage, directly or indirectly, caused by your use of such information. By accessing the Science Fair Project Ideas, you waive and renounce any claims against Education.com that arise thereof. In addition, your access to Education.com's website and Science Fair Project Ideas is covered by Education.com's Privacy Policy and site Terms of Use, which include limitations on Education.com's liability.
Warning is hereby given that not all Project Ideas are appropriate for all individuals or in all circumstances. Implementation of any Science Project Idea should be undertaken only in appropriate settings and with appropriate parental or other supervision. Reading and following the safety precautions of all materials used in a project is the sole responsibility of each individual. For further information, consult your state's handbook of Science Safety.