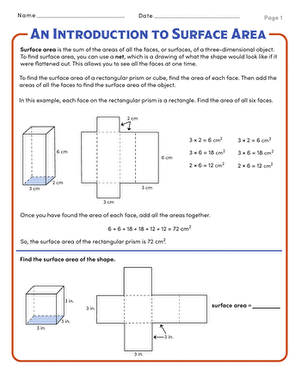
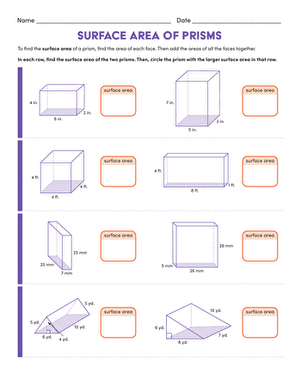
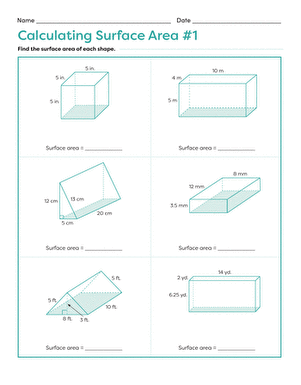
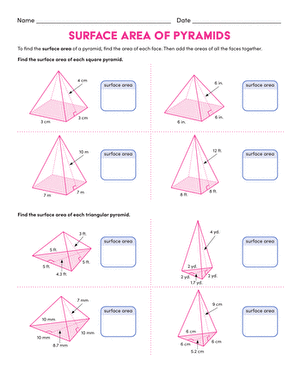
Activity
Soapy Surface Tension
Grade Level: 8th - 10th; Type: Physics
Objective:
Surface tension causes the molecules at the surface of a liquid (such as water) to cling together. This project examines the effects of soap on the surface tension of water.
Research Question:
How does soap affect the surface tension of water?
Did you know that the molecules on the surface of a cup of water stick together? This is because of a force called surface tension. In fact, if you carefully place a paper clip on the surface of a cup of water, it will seem to float because of surface tension. But what happens when a drop of soap hits the water’s skin? Find out with this science project.
Materials:
- Pennies
- Plastic Cup
- Water
- Eye dropper
- Liquid soap
Experimental Procedure:
- Fill a plastic cup with water.
- Dip the dropper into the cup, and suction in some water.
- Place drops of water from the dropper onto a penny, counting how many it takes for the water to spill off the penny. This is the number of drops it takes until the surface tension breaks.
- Dry off the penny.
- Repeat this process several times to make sure that you have an accurate count of how many drops it takes to break the surface tension.
- Dilute the water in the cup with a drop or two of liquid soap.
- Now see how many drops of water it takes until the water spills off the penny. How can you explain the difference?
Terms/Concepts: Surface tension; Water skidder (insect); How does soap interact with water?
References:
- Experiments With Bubbles, by Robert Gardner. Pp 18-20.
Education.com provides the Science Fair Project Ideas for informational purposes only. Education.com does not make any guarantee or representation regarding the Science Fair Project Ideas and is not responsible or liable for any loss or damage, directly or indirectly, caused by your use of such information. By accessing the Science Fair Project Ideas, you waive and renounce any claims against Education.com that arise thereof. In addition, your access to Education.com's website and Science Fair Project Ideas is covered by Education.com's Privacy Policy and site Terms of Use, which include limitations on Education.com's liability.
Warning is hereby given that not all Project Ideas are appropriate for all individuals or in all circumstances. Implementation of any Science Project Idea should be undertaken only in appropriate settings and with appropriate parental or other supervision. Reading and following the safety precautions of all materials used in a project is the sole responsibility of each individual. For further information, consult your state's handbook of Science Safety.