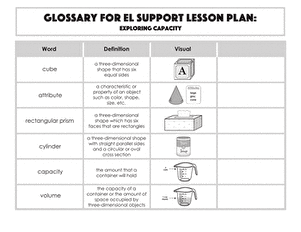
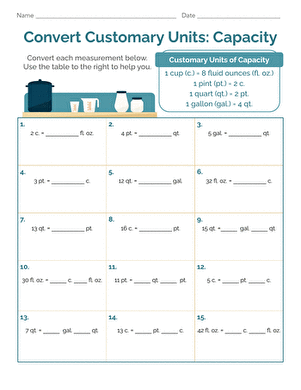
Activity
Heat Capacity of Water vs. Heat Capacity of Oil
Have you ever wondered why oil heats up so quickly in a pan, but water takes so long to boil?
Heat, which is the exchange of energy between a system and its surroundings, occurs in three major ways: conduction, convection and radiation.
Conduction is heat transfer through touch (physical contact between molecules). The hotter molecules are, the faster they move around and transfer their energy to other molecules. Convection is heat transfer through fluid flow, like when hot water is poured over ice or when cool air is blown over your warm soup. Radiation occurs when an object releases heat in the form of electromagnetic rays.
An object’s heat capacity describes the amount of heat required to change the temperature of that object by a certain amount. Specific heat is the amount of heat required to change the temperature of a substance by one degree (generally °C).
Liquids absorb heat in different ways. The temperature change in a particular liquid heated by conduction may not be the same trend of temperature change for the same liquid heated by radiation.
Problem
How do different liquids absorb heat?
Materials
- Water
- Salt water
- Olive oil
- Liquid soap
- Jars (however many liquids you have)
- Digital hot plate
- Digital thermometer
- Microwave
- Stopwatch
- Labeling tape
- Marker
- Any other liquid you want to test
Preparation
A night before you do your heat testing, measure a ½-cup of liquid into each jar and label it accordingly. You should have 2 jars for each liquid. Set the jars aside so they will all be the same temperature when you test them the next day.
Procedure
Microwave Testing
- Record the initial temperature of the liquid you are testing. Make sure to record your temperatures in °C.
- Place the jar with your first liquid in the microwave and heat on full power for 30 seconds. Record the temperature and any observations.
- Repeat step 2 several more times, recording the temperature and any observations each time. Be careful, the glass jar will get hot! Ask an adult to help you remove the jar from the microwave.
- Repeat steps 1-3 for your second liquid.
Hot Plate Testing
- Set the hotplate to 80°C.
- Record the initial temperature of the liquid you are testing.
- Place the jar on the hot plate and start the stopwatch.
- Record the temperature of the liquid every 2 minutes for 20 minutes. Record any observations.
- Be careful of the hot glass and liquid!
- Repeat steps 1-5 for your second liquid.
Plot Your Data:
Graph each set of data with temperature on the y-axis and time on the x-axis. What do your plots tell you?
Results
Microwaves are better at heating polar liquids, like water. Oils are very non-polar. Olive oil will heat up faster on the hotplate than water will. Water will still heat slower than olive oil when placed in the microwave, but your graphs should have indicated that water heats up faster in the microwave than it does on the hot plate.
Why?
For both the hot plate and the microwave, olive oil will heat up faster than water because the heat capacity of oil is lower than the heat capacity of water. Water requires more energy per gram of liquid to change its temperature. Because the input of the heat from the hotplate and the microwave is the same across trials, and water takes longer to heat up to a given temperature than olive oil, we can conclude the water can hold more heat energy than olive oil.
Microwave radiation is radiation with wavelengths of one millimeter up to one meter. These short wavelengths have high frequencies, and therefore high energies. Microwaves are much better at heating polar molecules, like water. A molecule is polar when it has a concentration of charge on one side or the other. On the water molecule, the oxygen atom is negatively charged, while the hydrogen atoms are positively charged. Oils, like olive oil, are long, evenly spaced chains and are very non-polar, so they don’t absorb energy from microwaves as well as polar molecules like water do. Microwaves work by causing the poles of a molecule to spin rapidly, which generates heat. This is known as dipole rotation.
Education.com provides the Science Fair Project Ideas for informational purposes only. Education.com does not make any guarantee or representation regarding the Science Fair Project Ideas and is not responsible or liable for any loss or damage, directly or indirectly, caused by your use of such information. By accessing the Science Fair Project Ideas, you waive and renounce any claims against Education.com that arise thereof. In addition, your access to Education.com's website and Science Fair Project Ideas is covered by Education.com's Privacy Policy and site Terms of Use, which include limitations on Education.com's liability.
Warning is hereby given that not all Project Ideas are appropriate for all individuals or in all circumstances. Implementation of any Science Project Idea should be undertaken only in appropriate settings and with appropriate parental or other supervision. Reading and following the safety precautions of all materials used in a project is the sole responsibility of each individual. For further information, consult your state's handbook of Science Safety.