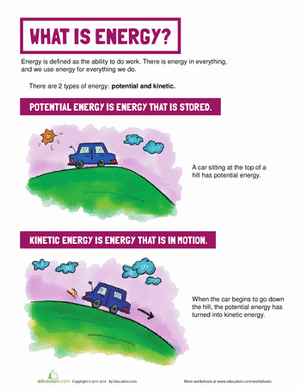
Activity
Electric Energy Drinks
Objective:
This experiment will explore if different energy drinks have different amounts of electrical energy.
Research Questions:
-Will different energy drinks power batteries with different levels of electricity?
-What ingredients in the energy drinks cause this difference?
Introduction:
Many Americans drink energy drinks for a quick burst of energy when they’re feeling tired. The next time you’re craving a can of this liquid fuel, remember that the same drink holds enough energy to power a battery! By transferring electrons between two electrodes, they can actually create electrical currents.
Materials:
- Energy drinks in different flavors and brands
- Water
- Jar
- Copper wire with insulation removed
- Zinc nail
- Voltmeter (find them at any hardware store)
- Sandpaper
- Journal
Experimental Procedure:
- Create a chart in your journal with the ingredients of each energy drink, noting any major differences.
- Fill the jar with water, and then clip one lead of the voltmeter to the wire and the other lead to the nail.
- Stick both the wire and nail into the jar, making sure they don’t touch.
- Record the reading on the voltmeter.
- Dump out the water, clean the jar and lightly sand and clean the wire and nail.
- Repeat steps 2 to 4 with each different energy drinks, and record your observations in your journal.
- Now it’s time to analyze your data. Which energy drink was the most electrically charged? Does there appear to be an ingredient that makes some drinks measure higher in charge than others? Could they affect humans?
Disclaimer and Safety Precautions
Education.com provides the Science Fair Project Ideas for informational purposes only. Education.com does not make any guarantee or representation regarding the Science Fair Project Ideas and is not responsible or liable for any loss or damage, directly or indirectly, caused by your use of such information. By accessing the Science Fair Project Ideas, you waive and renounce any claims against Education.com that arise thereof. In addition, your access to Education.com's website and Science Fair Project Ideas is covered by Education.com's Privacy Policy and site Terms of Use, which include limitations on Education.com's liability.
Warning is hereby given that not all Project Ideas are appropriate for all individuals or in all circumstances. Implementation of any Science Project Idea should be undertaken only in appropriate settings and with appropriate parental or other supervision. Reading and following the safety precautions of all materials used in a project is the sole responsibility of each individual. For further information, consult your state's handbook of Science Safety.
Education.com provides the Science Fair Project Ideas for informational purposes only. Education.com does not make any guarantee or representation regarding the Science Fair Project Ideas and is not responsible or liable for any loss or damage, directly or indirectly, caused by your use of such information. By accessing the Science Fair Project Ideas, you waive and renounce any claims against Education.com that arise thereof. In addition, your access to Education.com's website and Science Fair Project Ideas is covered by Education.com's Privacy Policy and site Terms of Use, which include limitations on Education.com's liability.
Warning is hereby given that not all Project Ideas are appropriate for all individuals or in all circumstances. Implementation of any Science Project Idea should be undertaken only in appropriate settings and with appropriate parental or other supervision. Reading and following the safety precautions of all materials used in a project is the sole responsibility of each individual. For further information, consult your state's handbook of Science Safety.